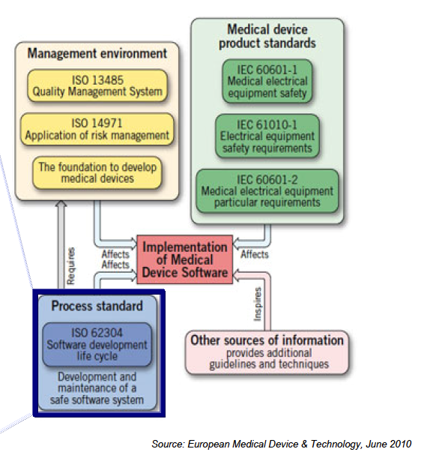
Prerequisites:
- ISO 62304:Quality Management System (QMS) – ISO 13485
- Risk management – ISO 14971
IEC 62304:
- Software safety classification
- Software development process
- Software management process
- Software configuration management process.
- Software problem resolution process
Medical Device Standards in One Frame